
Introduction
Modern biotechnology industries, such as those producing biopharmaceuticals, rely heavily on upstream bioprocessing where microorganisms and cells are used as biochemical 'factories'. Today the industry is managing three major challenges which can be summarized as compliance with increasing regulatory controls, cost control and risk reduction in drug development and manufacturing, and controlled deployment of new technologies.
An example of the industry's response to these challenges is the paradigm shift which is taking place in process development departments and 'upstream' bioprocessing. 'Downstream' typically describes the multiple steps in final preparation of the drug for distribution.
Historically, upstream processing has been performed using glass or stainless steel reactor vessels, termed bioreactors. In an effort to decrease the cost base, increase flexibility, and reduce development times, a change of operating model is underway employing single-use technologies, which require significantly less up-front investment, are highly modular, and faster to implement. The new bioreactors are either pre-sterilized disposable plastic bags or arrays of Tic-Tac boxes.
In parallel with the evolution of bioreactors the industry is seeking new methods and technologies to reduce process development times and increase process knowledge so that process efficiency and regulatory compliance is improved. Both FDA and EMEA initiatives such as Process Analytical Techniques (PAT) and Quality by Design (QbD) advocate the use of real-time, in-line monitoring of critical parameters in the manufacturing process. This presents opportunities for new control and measurement technologies to emerge. These new technologies must now be compatible with both the traditional and single-use bioreactors as must traditional control and measurement technologies.
This article describes once such technology: a solid-state, micro-optical sensor developed by Stratophase Ltd. that is suitable for both traditional and single-use bioreactors and provides users with a new process parameter to add to their armory for process control and monitoring.
Recent Advances In Bioprocessing
Today, bioprocess manufacturing is characterized by greater media complexity, lengthening process development times and an increasing rate of drug discovery. The recent history of bioreactors is dominated by either glass reactor vessels, which are typically used in process development and sized at 2 to 25 liters, or stainless steel, which are used in pilot plants and manufacturing. Stainless steel reactors can range from a few 100 to 10,000 liters or more. In the 1980's these were controlled mainly by off-line sampling and analog controls. By the 1990's, the vessels were very much the same, however, control had become digital. A number of the off-line measurements, for example, pH, had been converted to in-situ, real-time measurement.
Stainless steel manufacturing facilities require high capital investment, significant build and commissioning times and are inflexible to changes to output demand. They do have the advantage of re-usability but in many cases, this is more than offset by the cost of cleaning, sterilization and maintenance.
Single-use reactors for manufacturing are currently sized up to 2,000 liters, are faster to deploy, and require no on-going maintenance. As the industry moves to disposable reactors for manufacturing it is also seeking to reduce its process development costs and timescales. This is leading to the adoption of highly parallel mini-reactors, which can be less than 250ml and look similar to Tic-Tac boxes. These systems allow process development engineers to run multiple concurrent process trials as they seek to optimize their process.
Forecasted growth in single-use for manufacturing is expected to be 55% per annum over the next 3 years in a market valued at $2.8B by 2016. The paradigm shift from re-useable stainless and glass to single-use is a significant response to the industry's challenges.
Process Development, Control, And Measurement
These fundamental changes in both process development and manufacturing highlight a number of control and measurement (C&M) issues. C&M sensors now need to fit into much smaller reactor vessels. They need to be at a disposable price point and they must be usable across the entire range of reactor vessels, since glass and stainless steel will remain in use for some time. Some of the existing technologies may make the transition to this new operating model. Others may be more limited in their use.
As well as the physical requirement imposed on sensors by this paradigm shift, the industry is looking for sensors and systems which offer new measurement parameters to complement their existing techniques and allow users better process control and ultimately higher process yields and shorter development times. While real-time monitoring and control has been employed for many years in mature industries such as automotive and semiconductor, the biotechnology industry is heavily dependent on control strategies based on a combination of physical parameters such as pH, temperature and dissolved oxygen combined with off-line extractive sampling to verify the health of the media and, in particular, the status of the nutrient, typically glucose. Nutrient control is a critical parameter in determining the metabolic activity within the process and hence it's overall effectiveness.
While the use of off-line sampling systems is commonplace, the cost, both in consumables and in suitably qualified personnel is significant. As noted above, many Process Development departments are under pressure to reduce development times while product complexity is increasing. Their response is to increase their reactor capability with smaller, sub-1-liter vessels so that multiple process variants can be run in parallel. Extractive techniques are no longer viable at these small volumes since the sample represents a significant percentage of the total reactor volume.
It is also highly desirable to employ the same control and monitoring technology at all stages of the upstream process from process development through to manufacturing. Therefore, technologies which can be integrated into very small reactor vessels and single use bioreactors as well as traditional reactors are highly desirable.
Process Control And Feeding Strategy
Due to its simplicity and flexibility of use with a wide range of organisms and associated products, many bioprocesses rely on a fed-batch process for feeding organisms at the conditions required for optimum production. This is true with both fixed and single use models.
The majority of current fed-batch processes rely on the use of pre-determined feeding protocols based on nutrient requirement estimates, or the use of infrequent sampling and off-line assay of culture media in order to determine the concentration of key components. Both of these techniques can lead to non-optimum feeding, with depletion of nutrient and large swings in nutrient concentration risking the health of the organism and its ability to produce a high product titre.
An alternative to direct monitoring of nutrient concentration is to monitor the metabolic activity of the organism since this is a function of nutrient concentration. The majority of the organisms used in upstream bioprocesses, whether microbial or mammalian in nature, behave according to monod-type kinetics, which is valid for both exponential growth and steady-state culture under conditions of balanced growth.
The Monod relationship states that "as substrate concentration increases, the metabolic activity of an organism also increases towards a maximum" (see figure 1).
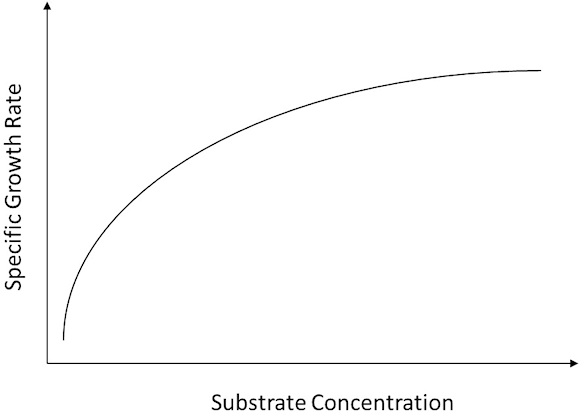
Fig. 1
As a result, as long as the substrate concentration is below that associated with the maximum, then relative nutrient concentration can be linked to an organism's metabolic rate. More specifically, characterizing the decrease in metabolic activity associated with decreasing nutrient concentration can be used as a trigger signal for on-demand feeding.
In order to take advantage of this effect a rapid and reliable indicator of metabolic activity is required. The monitoring technique needs to respond quickly to subtle changes in the metabolic rate in order to avoid the nutrient concentration decreasing to a point which is detrimental to the organism. In addition, the monitor ideally needs to respond directly to a metabolite or metabolites whose rate of concentration change is directly linked to the metabolic rate of the organism. The signal must also see minimal interference from changes associated with oxygen supply strategies or other bioreactor control parameters.
For one example, the Stratophase sensor technology and its associated control system offers such capability. The in-situ sensor (Ranger Probe) directly reports the process trend index (PTI) for the bioprocess being monitored, which is in effect a measure of the change in refractive index relative to the process. When the PTI is compared to the compositional changes that occur within a fed-batch process, a strong correlation between nutrient and/or product concentrations is observed.
The control system (Ranger Manager) analyzes the PTI in real-time to automatically calculate the metabolic rate (MRI) for the organism. Due to the PTI's direct response to changes in metabolite concentrations, the MRI is used to indicate metabolic activity and, by association, relative nutrient concentration. The control system uses the MRI to automatically trigger feed additions. When the MRI signal drops to a user defined pre-set value after the maximum has been passed a feed is triggered.
Each consumption cycle is independent from the previous, ensuring that feed addition is continually optimized for the organism at any given point in the process. The use of fixed volume feed additions results in control of nutrient concentration through variable frequency feed pulses. The Ranger Manager and Ranger Probe, when running automated closed loop feeding control, generate a real-time feeding regime which delivers an automatically optimized feed-on-demand dose of nutrient to the microorganism.
Technology
The innovative microptical sensor is based on solid state silicon and fibre optic technology and sits within the process reactor vessel (see figure 2). It can be deployed at all stages from process development to manufacturing and in glass, stainless and single use bioreactors.
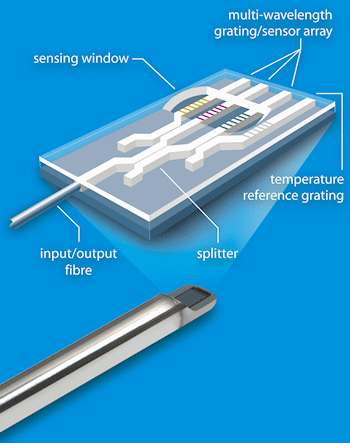
Fig. 2
The company's patented manufacturing process produces optical structures that route light around a small silicon chip and into the media. The light signal responds to compositional and metabolic rate changes within the reactor vessel in real-time. Stratophase has developed the first micro optical sensor which is truly optimized for real-time, in-situ bioprocess control and monitoring. This real-time measurement responds to the metabolic rate, enabling an optimized feeding regime for the bioprocess.
Product Integration
The Ranger System is composed of two components: Ranger Manager that controls the probe and a real-time sensor that sits within the process media (see figure 3). Communication between the two devices is by noise immune fiber-optic cable.
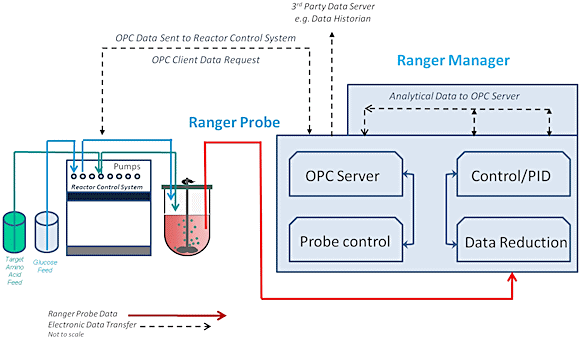
Fig. 3
Manager contains a laser light source, detectors, data reduction components, communication modules, and user interface components. The system features standard outputs such as OPC and 4-40mA to enable communication to process control systems.
Conclusion
The biotechnology industry is implementing significant changes in the type and usage of bioreactor vessels. The deployment of single use and highly parallel systems is advancing rapidly. This presents both a challenge and an opportunity for new technologies to emerge, which are useable in all bioreactor form factors. In addition, increasing complexity, higher value products and regulatory directives demand advances be made in process control.
Processing equipment is leading the way with smaller, massively parallel reactors and single use systems. Control and measurement technologies must advance rapidly to track these trends and offer new capability, which directly contribute to the profitability and cost effectiveness of the industry.
About the Author
Prior to joining Stratophase, Bill Campbell was CEO at Splashpower, a wireless power technology company, and Microemissive Displays (MED), a developer of P-OLED micro displays. He led the commercial development of both companies, through IP licensing in the case of Splashpower and through product development in the case of MED.
Related Content
Global Biometrics Technology Market Expected to Exceed USD 23 Billion by 2019
Wearable vapor sensor can 'smell' diabetes
LeapBand Wearable Activity Tracker For Children, Now Available
Sensor Technology Automates Feeding Control in Single Use Bioreactors
A Novel Micro-optical Sensor Automates Control of Bioprocesses